If you have read the AMH article in this series, you already know that AMH measures quantity – the number of eggs left in your reserve – not quality.
Quality is however what most people are actually worried about. And yet it is the less well understood of the two.
Egg quality, in the way clinicians use the term, refers primarily to chromosomal integrity – whether an egg contains the correct number of chromosomes. The age-related decline in that accuracy is real and significant. I am going to say that clearly here, and return to it properly at the end.
But chromosomal integrity isn’t the whole story. Eggs mature over months inside follicles – and the quality of that environment matters. The mitochondrial health of the egg. The oxidative burden it is exposed to. The hormonal conditions driving its development. The blood supply reaching the ovary. These are not fixed. They respond to how you live, what you eat and what you address.
That is what this article is about. Not a supplement list. An honest, evidence-based account – across nutritional medicine, lifestyle medicine, metabolic medicine and Traditional Chinese Medicine – of what the follicular environment needs, and what the research says about how to support it. Mechanism first, because in my experience, understanding why something works is what allows you to decide whether it is right for you.
What egg quality actually means – and where you can influence it
Every egg, at the point of fertilisation, should contain exactly 23 chromosomes. The process that achieves this – called meiosis – begins before birth but is not completed until the egg is triggered to ovulate, sometimes decades later.
The cellular machinery responsible for pulling chromosomes apart correctly during that final division is called spindle apparatus. In older eggs, this machinery becomes less precise. Chromosomal errors – known as aneuploidy – become more common. This is the primary driver of age related decline in egg quality, and is not fully reversible.
But egg quality is also shaped by a second layer – the cellular environment in which eggs develop:
- Mitochondrial function – how efficiently the egg generates the energy needed for cell division and early embryo development
- Oxidative burden – the degree of free radical damage the egg is exposed to as it matures
- Hormonal environment – the quality of signals driving follicular development
- Blood supply to the ovary – which determines the nutrients and oxygen reaching the follicle
These variables are responsive. The effect sizes are likely modest relative to the age effect – it is important to state that clearly. But removing avoidable obstacles is always worth doing. And that second layer is where meaningful influence lives.
The follicle is not just a container
An egg does not develop in days. The follicle that will eventually ovulate begins its development approximately three to four months before ovulation – a process called folliculogenesis. During those months, the egg is entirely dependent on its environment: The follicular fluid surrounding it, the granulosa cells lining the follicle and communicating directly with the egg, and the blood supply delivering nutrients, oxygen, and hormones into that fluid.
Follicular fluid is metabolically active. Studies measuring its antioxidant capacity, CoQ10 levels, fatty acid composition, and inflammatory markers have found consistent associations between a healthier follicular environment and better oocyte and embryo outcomes.
The practical implication of this is significant – and worth sitting with.

Energy: The Mitochondrial Story
Eggs are the most mitochondria rich cells in the human body. Most cells contain a few hundred mitochondria. A mature egg contains several hundred thousand.
The reason is energy demand. Completing the final stages of meiosis, being fertilised, sustaining the earliest cell divisions of the embryo – before the embryo’s own genome activates – all require enormous amounts of ATP, generated by mitochondria. When mitochondrial function is compromised, the spindle apparatus becomes even less precise. Chromosomal segregation errors increase. The early embryo has less to work with.
Mitochondrial function declines with age. It is also influenced by nutrition – specifically, by the availability of the compounds mitochondria need to run efficiently.
CoQ10/Ubiquinol
Coenzyme Q10 sits at the heart of the mitochondrial electron transport chain – the mechanism by which ATP is produced. Without adequate CoQ10, the chain runs inefficiently and generates more damaging free radicals as a byproduct. CoQ10 levels in ovarian tissue decline with age.
The most cited study here is Bentov et al. (2013), which showed that CoQ10 supplementation in aged mice improved mitochondrial function in eggs, reduced aneuploidy rates, and improved reproductive outcomes. Human data is more limited but consistent in that direction: a randomised controlled trial by Xu et al. (2018) in poor IVF responders found improved ovarian response and embryo quality with CoQ10 versus placebo.
A note on form and dose: CoQ10 comes as ubiquinone (oxidised) or ubiquinol (the reduced, active form). Ubiquinol is better absorbed, particularly as conversion capacity declines with age. Doses in fertility research range from 200mg to 600mg daily.
The mechanistic rationale is strong. The safety profile is excellent.
Oxidative stress: protecting the environment
Oxidative stress occurs when free radicals exceed the body’s capacity to neutralise them. The follicular environment is particularly vulnerable – and its antioxidant adequacy has been directly correlated with oocyte and embryo quality in multiple studies.
The mechanism is direct. Free radicals can damage the spindle fibres responsible for chromosomal segregation – compounding, not replacing, the age-related effect. A 2022 systematic review (Barbagallo et al.) found that higher antioxidant capacity in follicular fluid was consistently associated with better oocyte quality and embryo development.
Diet is the most evidence based lever here. The Mediterranean dietary pattern – vegetables, legumes, whole grains, olive oil, oily fish, colourful fruit – addresses antioxidant status, omega-3 adequacy, and anti-inflammatory balance simultaneously. A 2018 prospective cohort study (Karayiannis et al.) in women undergoing IVF found that higher adherence to a Mediterranean diet was associated with significantly higher rates of clinical pregnancy and live birth.
Specific nutrients with antioxidant roles in follicular fluid include vitamin C (found at high concentrations relative to plasma), vitamin E, alpha lipoic acid, and N-acetyl cysteine – a precursor to glutathione, the body’s primary intracellular antioxidant. Targeted supplementation is used in clinical fertility protocols, though dietary foundation comes first.

Inflammation: the quiet disrupter
Chronic low-grade inflammation disrupts follicular development – quietly, without obvious symptoms, but consistently. Pro-inflammatory cytokines detected in follicular fluid are associated with poorer oocyte quality and lower fertilisation rates. Inflammation also drives oxidative stress, which compounds its effects.
The most common drivers are familiar: a diet high in ultra-processed foods, poor gut health, disrupted sleep, chronic stress, excess visceral adiposity. None of these are fixed. All of them are addressable.
Omega-3 fatty acids deserve specific mention. EPA and DHA – found in oily fish – have direct anti-inflammatory effects and are incorporated into cell membranes, including those of eggs and granulosa cells. DHA has been found at relatively high concentrations in follicular fluid. A 2019 meta-analysis (Arias-Alvarez et al.) found that omega-3 supplementation was associated with improved IVF outcomes across multiple markers.
Two to three portions of oily fish per week – salmon, mackerel, sardines, anchovies – or a high quality fish oil or algae-derived supplement at 1-3g combined EPA/DHA daily, is a well supported approach.
Three hormonal variables worth checking
Insulin, thyroid and cortisol each have direct, well-evidenced effects on follicular development. They are not fringe concerns – they are mainstream physiology, and they are not always adequately assessed in standard fertility care.
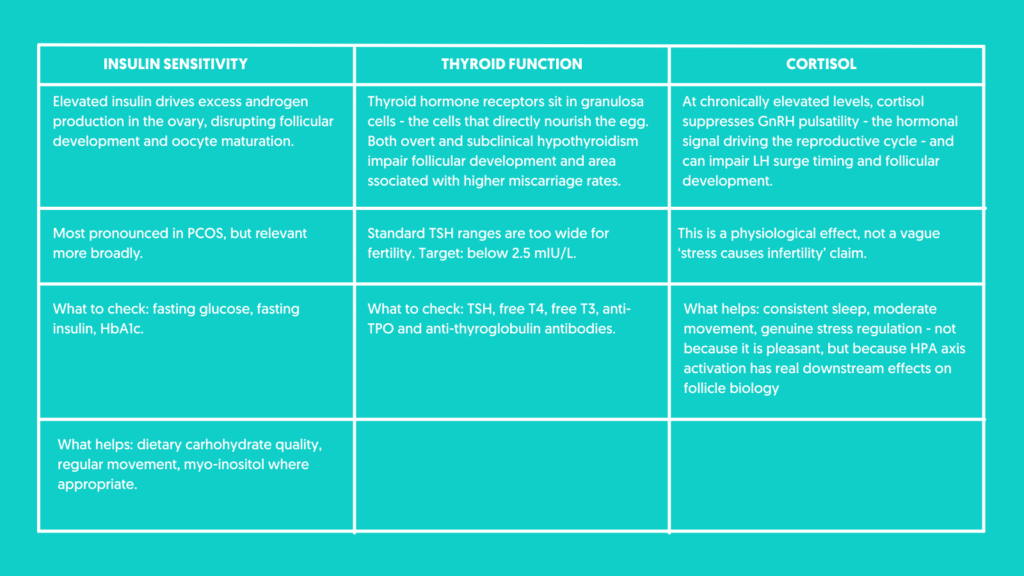
Blood flow and Traditional Chinese Medicine
Ovarian blood flow does not feature prominently in Western fertility consultations. It should.
The quality of follicular fluid – and the environment in which eggs develop – is directly dependent on blood supply. Blood carries oxygen, nutrients, hormones, and the raw materials the follicle needs. Reduced ovarian blood flow means a compromised follicular environment, regardless of what is being taken orally.
This is where Traditional Chinese Medicine enters the conversation – and where the evidence is more interesting than mainstream medicine often acknowledges.
Acupuncture’s proposed mechanisms include modulation of the autonomic nervous system – reducing sympathetic tone, increasing parasympathetic activity – which has direct effects on vascular smooth muscle and blood flow. Several studies using Doppler ultrasound have demonstrated increased uterine and ovarian blood flow following acupuncture. A 2020 systematic review (Smith et al.) found evidence of improved ovarian blood flow parameters in women with diminished ovarian reserve, alongside improvements in AMH and antral follicle count in some studies.
Acupuncture also influences the HPA axis – reducing cortisol and modulating stress hormones – and has been studied for its effects on FSH levels and follicular development. The evidence is not yet at the level of large-scale, well-controlled RCT, but the mechanistic plausibility is real, the safety profile is excellent, and the body of evidence is more substantial than dismissive positions often acknowledge.

Sleep: the most underrated Variable in this picture
Most people know melatonin as the hormone that regulates the sleep-wake cycle. What is less widely known is that melatonin is also produced locally within the follicle – by the granulosa cells surrounding the developing egg – where it acts as one of the most potent antioxidants in the follicular environment.
Melatonin concentrations in follicular fluid are significantly higher than in blood plasma. A landmark study by Tamura et al. (2012) demonstrated that melatonin in follicular fluid directly scavenges free radicals, protecting the egg from oxidative damage during the critical period of final maturation. Women with higher levels had better fertilisation rates and embryo quality.
Subsequent RCTs have explored melatonin supplementation in IVF, finding improvements in oocyte quality and fertilisation rates in poor responders. Melatonin – typically 3mg at night – is now used in some fertility clinic protocols.
Melatonin production is suppressed by light, particularly blue light in the hours before sleep. Chronic sleep restriction and disrupted sleep will result in lower melatonin levels. Poor sleep also independently elevates inflammatory markers and cortisol, compounding its effects on the follicular environment.
What cannot be changed – said honestly
The primary driver of declining egg quality over forty is the age related deterioration of the spindle apparatus. It is driven by accumulated oxidative damage and the gradual degradation of cellular machinery set in place before birth.
No supplement, protocol, or lifestyle change will restore chromosomal accuracy in older eggs to the level of younger ones. The aneuploidy rates at 40, 42, 44 are what they are.
But, please remember – they describe a population, not an individual. Healthy live birth rates occur across all of these ages. But honest medicine requires naming the primary constraint.
My take on the evidence
The women most likely to benefit from a sustained, evidence-based approach to egg quality support are those with identifiable, addressable factors – suboptimal thyroid function, poor sleep, a diet low in antioxidants and omega-3s, insulin sensitivity, or high chronic stress.
Addressing those factors is not speculative. It is removing avoidable obstacles from a picture that already has enough unavoidable ones.
The honest answer to ‘will this work?’ is: it may improve the conditions in which your eggs are developing. It will not override the age effect. Both of those things are true – and neither cancels the other out.
Where to start: A framework, not a protocol
This is not a checklist. I’m offering it as a way of thinking about what to prioritise – and in what order – so you can make an informed choice about what you do next.

And finally, give it time. A minimum of three months, ideally longer. The follicular development window means that changes made today have their most meaningful effect on eggs developing now.
Start where you are.
References & Evidence Notes
Bentov Y et al. The use of mitochondrial nutrients to improve the outcome of infertility treatment in older patients. Fertility and Sterility. 2013.
Xu Y et al. Coenzyme Q10 improves ovarian response and embryo quality in low-prognosis young women with decreased ovarian reserve: a randomized controlled trial. Reproductive BioMedicine Online. 2018.
Florou P et al. Does coenzyme Q10 supplementation improve fertility outcomes in women undergoing assisted reproductive technology procedures? Journal of Assisted Reproduction and Genetics. 2020.
Barbagallo F et al. Systematic review: follicular fluid antioxidant capacity and oocyte quality. Antioxidants. 2022.
Karayiannis D et al. Adherence to the Mediterranean diet and IVF success rate among non-obese women attempting fertility. Human Reproduction. 2018.
Arias-Alvarez M et al. Omega-3 fatty acids and IVF outcomes: a meta-analysis. Reproductive Biology and Endocrinology. 2019.
Smith CA et al. Acupuncture for women undergoing IVF: updated systematic review and meta-analysis. Journal of Clinical Medicine. 2020.
Tamura H et al. Melatonin and female reproduction. Journal of Obstetrics and Gynaecology Research. 2014.
Espino J et al. Impact of melatonin supplementation in women with unexplained infertility undergoing fertility treatment. Antioxidants. 2019.
NICE Clinical Guideline CG156: Fertility Problems — Assessment and Treatment. 2013, updated 2017. https://www.nice.org.uk/guidance/cg156
Leave a Reply

READ >
If you have read the AMH article in this series, you already know that AMH measures quantity – the number of eggs left in your reserve – not quality. Quality is however what most people are actually worried about. And yet it is the less well understood of the two. Egg quality, in the way […]

READ >
You got a number. Maybe a doctor read it out to you at the end of a ten-minute appointment and moved swiftly on. Maybe you ordered a home test, opened an app, and found yourself staring at a figure you didn’t know how to hold. Maybe you’ve known your result for months and have spent […]

READ >
Dr Deepak Ravindran is one of the UK’s leading pain specialists. He is one of the few medical consultants who possesses triple certification in lifestyle medicine, musculoskeletal medicine and pain medicine using all of this knowledge to give his patients a truly integrative and holistic approach. He is author of the book The Pain-Free Mindset […]

READ >
Ellie Grace is a trauma informed yoga educator with an MA in Yoga Studies – who lectures medical undergraduates in the neurophysiology of yoga to nurture their personal development and prevent burnout – which has never been more important. Doctors are experiencing workplace burnout at the highest ever levels recorded, with findings from the regulator’s […]

READ >
Lisa Ryan is a physiotherapist turned trauma informed somatic practitioner who helps people find freedom from their persistent pain. And this is just so important to her – because she was once a patient herself who was told she was not expected to recover – and the best she could hope for was to manage […]

READ >
Tanya Cole-Lesnick is a psychotherapist, licensed clinical social worker, and personal development coach who is focused on helping people clear their energetic clutter to pursue a life that honors their truest selves. Tanya really believes in the power of the group in the work she does – which is what I want to talk to […]